
Ames Procedural
Requirements
APR 1700.1
COMPLIANCE IS MANDATORY
Ames Health and Safety Manual
15.4.1 Supervisors
15.4.2 Safety, Health and Medical Services Division
15.4.3 Employees
15.5.1 Personal Protective Equipment
15.5.2 Containers and Equipment
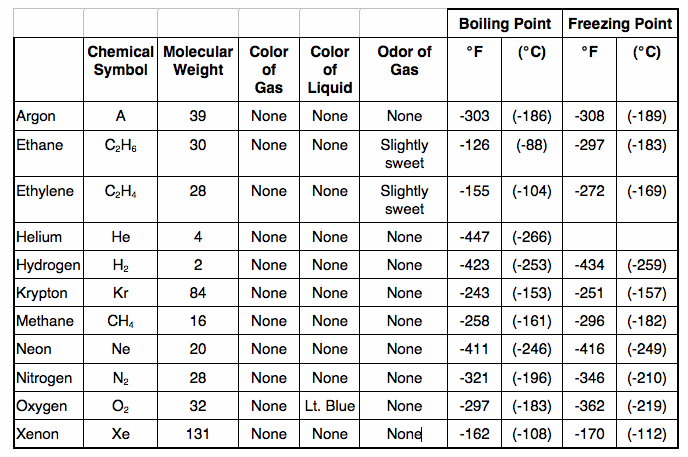
Appendix A: Physical Properties of Low-Temperature Liquefiable Gases
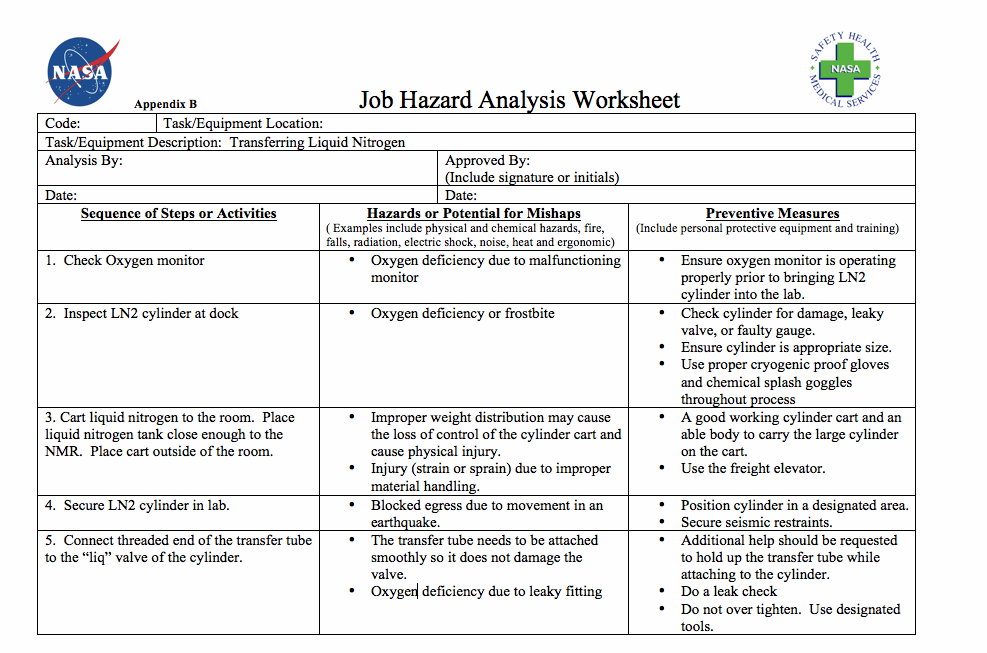
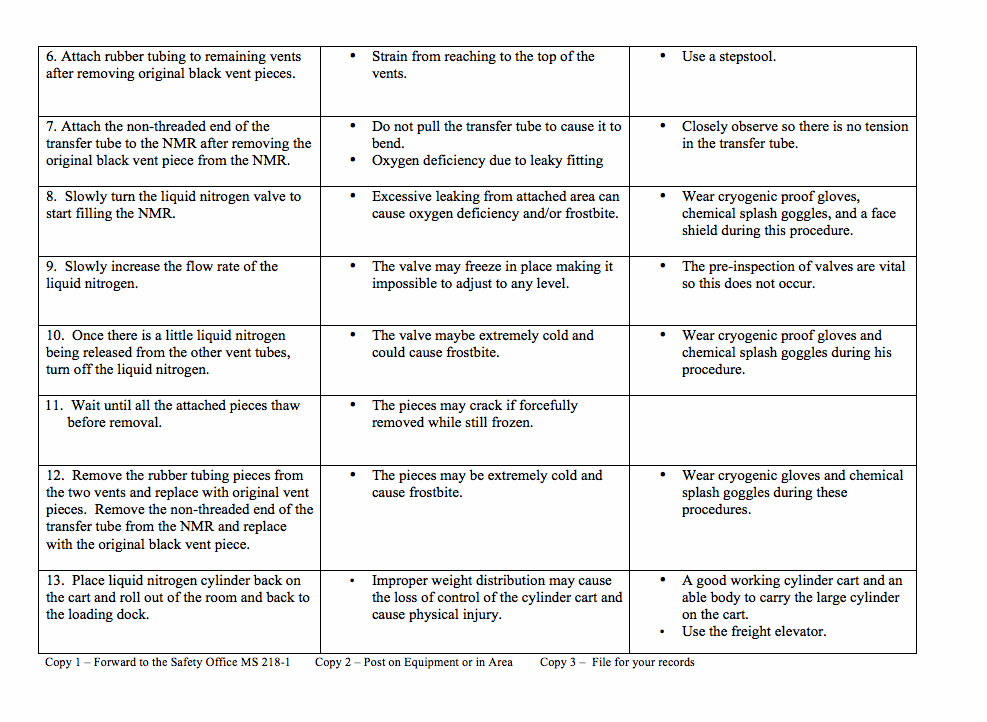
Appendix B: Job Analysis Worksheet
Appendix C: Safety Rules for Handling Cryogens
This chapter is to provide safety information and requirements for cryogenic materials.
[Back to Top] [Back to Main Table of Contents]
[Back to Top] [Back to Main Table of Contents]
Cryogenic Liquids - A liquid with a normal boiling point below- 130 degrees F
Dewars - Insulated storage tanks designed specifically to store cryogenic fluids
Pressure Relief Devices - Used to prevent pressure buildup in a system if it exceeds a pre-determinated amount.
[Back to Top] [Back to Main Table of Contents]
[Back to Top] [Back to Main Table of Contents]
The minimum required PPR for handling anything that is, or may have been, in contact with croygenic materials includes:
Cryogenic dewars shall not be accompanied by the personnel when transported by elevator. An oxygen deficient atmosphere could occur if The cryogen released in the elevator.
[Back to Top] [Back to Main Table of Contents]
All permanent oxygen detection systems must be supervised by NASA Dispatch.
A supervised system may be allowed only with review and approval of the Safety, Health and Medical Services Division.
[Back to Top] [Back to Main Table of Contents]
Follow all requirements in the NASA NSS 1740.16 (Hydrogen) and NSS 1740.15 (Oxygen).
[Back to Top] [Back to Main Table of Contents]
[Back to Top] [Back to Main Table of Contents]
Cyrogenics is a field that uses or studies materials at extremely low temperatures. According to the National Institute of Standards and Technology, the term applies to any work conducted at temperatures lower than -150 degrees Centigrade (-238 degrees Fahrenheit).
Cryogenics uses gases that liquify at extremely cold temperatures, including nitrogen, oxygen and helium.Cryogenic liquids require cautious handling. The liquid gases can make carbon steel, plastics and other high-strength materials fracture or shatter with ease. Contact with eyes or the skin can cause serious frostbite injuries. Flesh sticks to super-cooled materials, even nonmetallic materials, and trying to remove skin stuck to any frozen material can tear flesh.
Skin exposed to a cryogenic liquid or gas should be restored to normal body temperature by running warm liquid over it. The warming liquid should never exceed 112 degrees Fahrenheit and the affected body parts should never be rubbed. Victims should get emergency care as quickly as possible.
A less obvious danger from cryogenic liquids comes when they warm and while reverting to a gaseous state, displace oxygen. Breathing oxygen-deficient air can cause dizziness, nausea, vomiting, unconsciousness and confusion. Depending on how rapidly the gas expands, unconsciousness can occur without being preceded by any of these warning symptoms. For this reason, cryogenic liquids should always be used in open, well-ventilated areas. If that is not possible, use air monitoring devices to measure oxygen levels and signal when oxygen drops to dangerous levels.
An oxygen-supplying respirator, such as an airline respirator with an egress bottle or a self-contained breathing apparatus (SCBA) is essential when working in an oxygen-deficient atmosphere. Cartridge-style respirators will not help. Persons overcome by loss of oxygen should be moved to a well-ventilated area immediately. Rescuers may need an SCBA to avoid falling victim themselves. If breathing is difficult, oxygen should be supplied. If breathing has stopped, artificial respiration should be administered.
While transferring and handling cryogenic liquids, always wear chemical splash goggles and face shields. Wear gloves made to withstand extremely low temperatures. The gloves should fit loosely so they can be thrown off quickly. Wear long-sleeve shirts and trousers to protect all parts of the skin. Pant legs should go over the tops of footwear so spills cannot get into boots or shoes.
When cryogenic liquids warm up, the huge amounts of gas they are capable of producing can cause explosions or vessel ruptures. For this reason, the liquids should never be kept in plugged or covered containers. Use containers made specifically for cryogenic liquids, such as Dewar flasks. These consist of one flask inside another with a space in between providing insulation. The insulation keeps the liquid from warming quickly and causing rapid gas expansion. Even though Dewar flasks are made to withstand the stress of extreme temperature changes, they should be filled very slowly to protect the containers from excessive internal stress. They should not be filled past 80 percent of their capacity to allow for gas expansion.
Because cryogenic liquids can boil at room temperatures, causing eruptions and splashes, tongs should be used to remove anything immersed in the liquid. For more information, see EZ Facts� document No. 290, How to Work Safely with Cryogenics. Go to: http://www.labsafety.com/refinfo/ezfacts/ezf290.htm. For a selection of cryo-related products, go to: http://www.labsafety.com/search/results.asp?prodKeyword=Cryo.
[Back to Top] [Back to Main Table of Contents]
Liquid, vapor, or low-temperature gas will produce effects on the skin similar to a burn. This will vary with temperature and exposure time. Vapors issuing from cryogenic liquids can damage tissue including the eyes, which can be damaged by an exposure to cold gases too brief to affect the skin.
[Back to Top] [Back to Main Table of Contents]
![]()